Palladium‐Catalyzed Direct Arylation of (Hetero)Arenes with Aryl Boronic Acids - Yang - 2008 - Angewandte Chemie International Edition - Wiley Online Library

Binuclear Palladium Complex Immobilized on Mesoporous SBA-16: Efficient Heterogeneous Catalyst for the Carbonylative Suzuki Coupling Reaction of Aryl Iodides and Arylboronic Acids Using Cr(CO)6 as Carbonyl Source | SpringerLink

Fluorinated Aryl Boronates as Building Blocks in Organic Synthesis - Budiman - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library
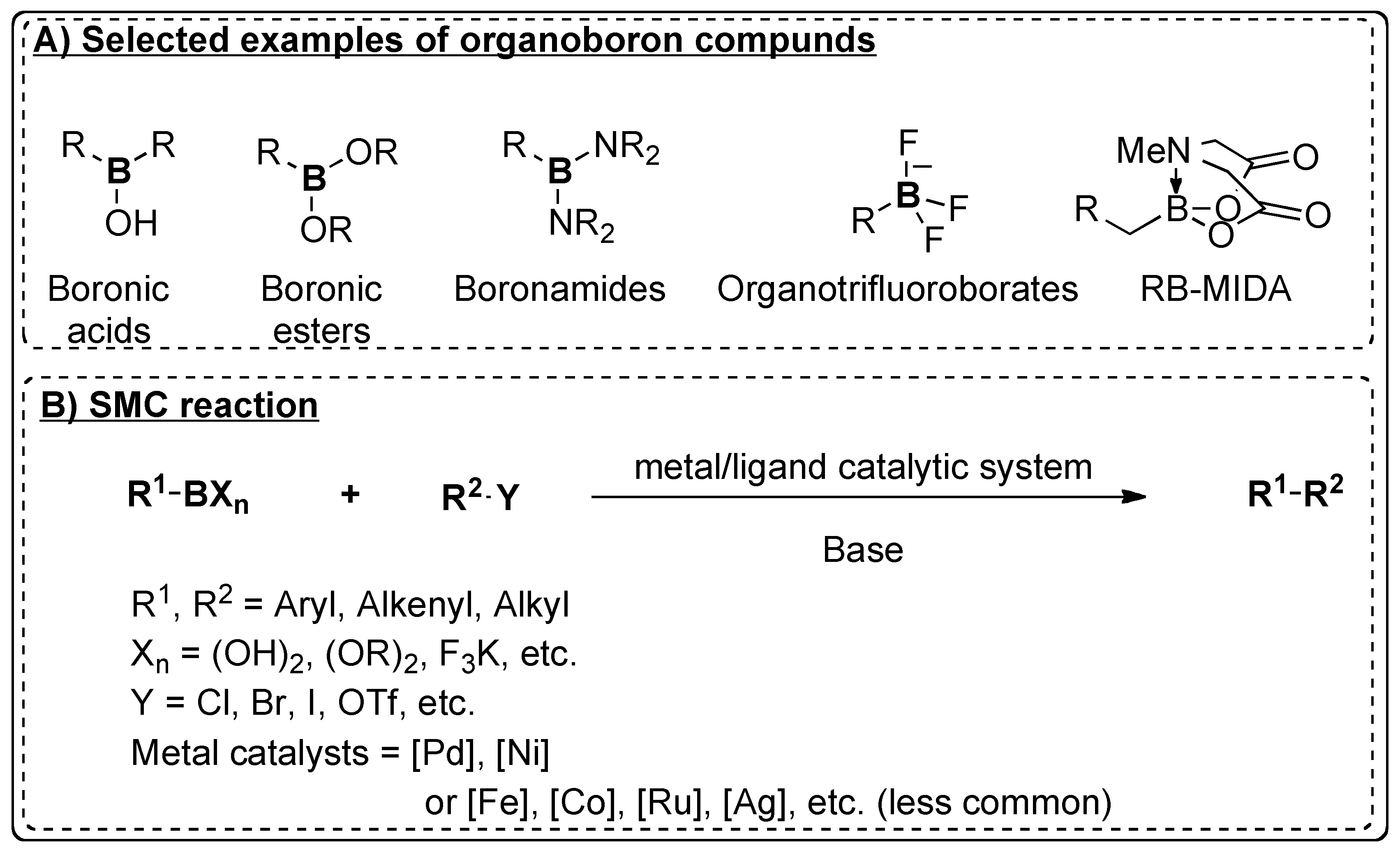
Catalysts | Free Full-Text | Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings | HTML

Scope of the palladium-catalyzed aryl borylation utilizing bis-boronic acid. - Abstract - Europe PMC

Suzuki–Miyaura Cross‐Coupling Reactions of Alkylboronic Acid Derivatives or Alkyltrifluoroborates with Aryl, Alkenyl or Alkyl Halides and Triflates - Doucet - 2008 - European Journal of Organic Chemistry - Wiley Online Library
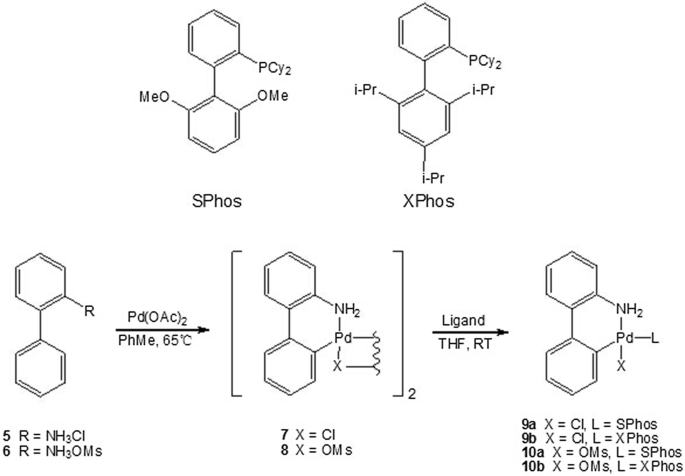
Palladium-catalyzed borylation of aryl (pseudo)halides and its applications in biaryl synthesis | BMC Chemistry | Full Text

Palladium-catalyzed, direct boronic acid synthesis from aryl chlorides: a simplified route to diverse boronate ester derivatives. | Semantic Scholar

Palladium-Catalyzed Methylation of Aryl, Heteroaryl, and Vinyl Boronate Esters. - Abstract - Europe PMC

Suzuki-Miyaura cross-coupling reaction of aryl chlorides with aryl boronic acids catalyzed by a palladium dichloride adduct of N-diphenylphosphanyl-2-aminopyridine - ScienceDirect

Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids - ScienceDirect
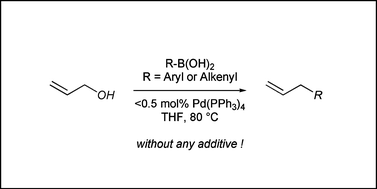
Palladium(0)-catalyzed direct cross-coupling reaction of allylic alcohols with aryl- and alkenylboronic acids - Organic & Biomolecular Chemistry (RSC Publishing)

Table 2 from A palladium-catalyzed three-component cross-coupling of conjugated dienes or terminal alkenes with vinyl triflates and boronic acids. | Semantic Scholar

Highly enantioselective rhodium-catalyzed cross-coupling of boronic acids and racemic allyl halides | Nature Protocols